01
Aspyre probes hybridize to both healthy and mutated DNA but are mismatched to healthy molecules.
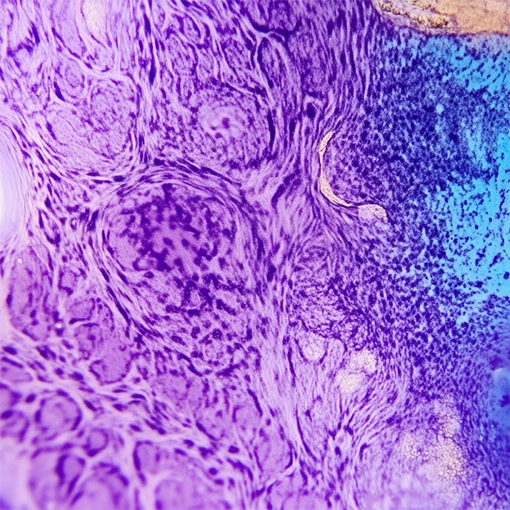
Targeted cancer therapies have dramatically improved survival rates1, but only patients who receive biomarker testing can benefit from them. And there are critical barriers to these tests throughout the patient journey.
Existing biomarker tests, such as next generation sequencing (NGS), are complex and take weeks to deliver results. Many samples fail, sometimes requiring repeat or liquid biopsies which may cause additional delays.

An alternative to NGS enabling simplified genomic profiling, Aspyre solves the challenges of existing technologies and makes it easier to get actionable biomarker information. Aspyre is designed to run on existing PCR instruments, of which there are more than 100,000 globally, and comprises just four steps. Aspyre also includes turnkey data analysis that is easy to implement, enabling more patients to get the right treatment as soon as possible after diagnosis.
Aspyre probes hybridize to both healthy and mutated DNA but are mismatched to healthy molecules.
A DNA polymerase specifically digests only perfectly matched probes. Those annealed to healthy DNA are not digested due to mismatch.
Digested probes are selectively circularized by ligation.
Only circularized probes are amplified and detected. Probes that hybridized to healthy molecules are not amplified.
The first clinical application of the Aspyre technology, Aspyre Clinical Test for Lung provides fast, actionable biomarker data for non-small cell lung cancer (NSCLC) patients.